A Shot at Cancer
Both had the blessings of Dr. Richard Klausner, then NCI director. But even Klausner, a well-respected researcher, had to be persuaded at first. "I came to the NCI being quite skeptical about it," he says of the new strategy.
And he wasn't the only one. What Schwartzentruber and Kwak were hoping to do was prove they could vaccinate a patient against cancer — educate a body to, in essence, recognize and round up tumor cells the same way it polices viruses and bacteria. It certainly made good biological sense: the immune system is the body's built-in defense mechanism, after all, so why not turn it against one of the most ornery diseases around?
The problem, of course, is that a tumor is not exactly a pathogen. What it is, at its core, is a collection of aggressively growing cells that can't stop dividing. It is not entirely foreign, as a virus is; it does not infect healthy cells, as bacteria and viruses do. Turning the immune system against cancer cells would involve turning the body's defense mechanisms against a part of itself. Designing a vaccine to do this entails creating the biological version of a stealth weapon encased in a smart bomb equipped with a guided missile.
And that was proving to be a bit too challenging. Nothing that hundreds of researchers in hundreds of trials had attempted had worked. While the vaccine idea made logical sense, the immune system, it seemed, just wasn't designed to battle cancer this way.
But in June, after nearly a decade of carefully inoculating patients suffering from either advanced melanoma or a type of lymphoma, both Schwartzentruber and Kwak announced positive outcomes of their trials, at the American Society of Clinical Oncology meeting in Orlando, Fla. Their results, along with those of a trial vaccine against prostate cancer and an early candidate against a type of brain cancer, suggest that we might finally be on the way to unleashing the immune system against the disease.
It's about time. Senator Edward Kennedy's death after a yearlong battle with brain cancer is only the most prominent reminder that while many current treatments are certainly effective, they can be made even better. Though malignancies are now being caught earlier than ever before and treatments that target and control the disease are more effective than ever before, cancer is still the second biggest killer in the U.S., claiming more than half a million lives each year. Surgery, chemotherapy and radiation can do only so much when tumor cells hide in plain sight and even a single overlooked cell can seed new disease.
That's where a vaccine-based strategy could make a difference. An immune system trained to recognize the first signs of new or recurrent growth can begin to attack malignancies far earlier than the best scans can detect them. And the latest vaccines incorporate clever new insights into how malignant cells can be tagged, exposed and destroyed. "Understanding how the immune system works is going to play a significant role in our treatment of cancer going forward," says Dr. Len Lichtenfeld of the American Cancer Society.
It's not just the biology that is getting better. Researchers are even fine-tuning when to give a cancer vaccine. The latest data from the lymphoma trial, for example, suggest that in some cases, the best time to train the immune system might be during a remission, when the body's defensive cells are at their strongest. "It's been a slow evolution, but we are seeing the first inklings that cancer vaccines can work," says Dr. Steven Rosenberg, chief of the surgery branch at NCI and a cancer-vaccine pioneer who trained Schwartzentruber and Kwak.
When Is a Vaccine Not a Vaccine?
There may be no better example of what is meant by preventive medicine than the strategy of vaccination. A healthy person is given a tiny taste of a virus — flu or polio, say — that's too weak to cause illness but just enough to introduce the body to the pathogen. If the virus later shows up for real, the immune system is primed and waiting for it.
That's close to how a cancer vaccine works, but not precisely. Most experts see cancer vaccines as a hybrid of treatment and prevention. While it's true that the Food and Drug Administration has approved vaccines against cervical and liver cancer, both are actually designed to fight the viruses most responsible for causing the disease, as opposed to targeting cancer itself — human papillomavirus in the case of cervical cancer and hepatitis B in the case of liver tumors.
Using vaccines to prevent nonviral cancers in someone who is disease-free is a whole different matter. For one thing, it's much more difficult to determine a person's chance of developing a particular type of cancer than it is to determine the likelihood of being exposed to, say, the influenza virus or chicken pox. What passes for "exposure" in the case of nonviral cancers is a combination of genes and environment and a range of other X factors that can vary from person to person. How do you vaccinate against your family legacy of breast cancer or your constant exposure to secondhand cigarette smoke?
But that doesn't mean the immune system can't be exploited in a different way. Cancer vaccines would ideally be used in patients whose disease has already been diagnosed and treated with surgery, chemotherapy or radiation. They would then be immunized as a way to prevent the cancer from coming back and spreading. Such metastases are actually the leading cause of death from cancer. "The charm of working with the immune system is that we can use the body's own defense mechanisms to possibly get to that last cancer cell or at least create a surveillance system that keeps that cancer under control," says Lichtenfeld.
Trial by Failure
Before they can seek out these smaller, hidden deposits of tumors, however, cancer vaccines must prove that they can actually target and shrink a cancer's more conspicuous growths. This, it turns out, is obvious in theory but devilishly challenging to show in reality.
Take melanoma. In 2002 scientists at the John Wayne Cancer Institute in Santa Monica, Calif., thought they had finally figured out a way to turn the immune system against the skin cancer. Instead of trying to activate immune cells with snippets of tumor proteins they had created in the lab, they decided to grind up melanoma tumors and use the malignant slurry to prod the right immune cells into action. The result was Canvaxin, a vaccine against aggressive melanoma that was loaded up with 20 different tumor-specific components of melanoma, teaching the body new ways to recognize the disease. More than 1,500 patients were given the vaccine after being treated with surgery and chemotherapy. In the first five years of follow-up, the shot proved safe and worthy of moving into the most advanced level of human testing. But in April 2005, the scientists and the biotech company they had enlisted to develop the vaccine were forced to stop their studies when it became obvious that the vaccinated patients were not living any longer than the unvaccinated ones. "That put a damper on things," says Schwartzentruber. "They had what they thought was a promising start, and it was an international, multi-institutional study with a large number of patients."
In retrospect, Schwartzentruber says, the problem may have been that the vaccine was forced to work alone. Even the most well-sensitized immune system may be fooled by the homegrown nature of cancer, recognizing malignant cells as just another part of the body — which they are — and thus giving them a pass. When the cancer finally grows big enough to represent a real threat, it's too late.
Schwartzentruber thinks he has a way around that problem. In some trials, after giving his vaccine to patients with advanced melanoma that has spread to other tissues, he adds an immune stimulator called interleukin-2 (IL2) for reinforcement. Alone, the vaccine would not cause any tumors to shrink. The IL2 treatment itself wasn't very effective either; it shrank tumors in only 10% of patients. But combining the vaccine and IL2 has caused tumors in 22% of patients to regress — a doubling of effectiveness. "This teaches us a lesson: that combinations of biologic treatments are more powerful than their individual components," says Schwartzentruber.
Kwak and his collaborators, led by Dr. Stephen Schuster at the University of Pennsylvania, see a similar power in pairing. Their vaccine, against a form of non-Hodgkin's lymphoma known as follicular lymphoma, takes a slightly different, more personalized approach. Rather than relying on a commonly found antigen or snippet of cancer protein to teach the body to recognize the malignancy, they designed each vaccine using individual patients' specific lymphoma profiles. They then partnered this customized concoction with another immune stimulator, GMCSF. Patients receiving the combination remained in remission on average 44 months after the vaccination, a 47% improvement in disease-free survival compared with those getting the uncustomized vaccine, who stayed in remission for just 30 months.
This study is also the one that yielded the most evidence that the best time to inoculate patients is when they're in remission from their disease. While Schwartzentruber elected to administer his melanoma vaccine when his subjects were in the most advanced stages of illness, Kwak and his colleagues decided to capture the immune system at its best. They waited until the patients had been in remission for six months after chemotherapy, which rid the body of the bulk of the tumor burden. Give the immune system a break from that life-or-death battle, and it might be better able to do the surveillance work of corralling stray cells that escape the initial treatment. "I envision that vaccine approaches like this could be useful as maintenance therapy," says Kwak. "We would use chemotherapy and surgery to debulk the tumor and then vaccinate to maintain remission."
The Riddle of Success
Another way to make a vaccine more effective might be to manipulate the very nature of the tumor, so that it is a more obvious target for the immune system — a little like tying a more colorful fly on a fishing hook. The idea, says Dr. Patrick Hwu, chair of melanoma oncology at the University of Texas' M.D. Anderson Cancer Center and a member of Schwartzentruber's team, is to "get the tumor itself to look like a virally infected site, to get the whole immune system going."
The untreated immune system is not helpless in all of this. Rosenberg has biopsied tumors and extracted immune cells called lymphocytes from patients with advanced cancer and has grown these cells in culture. In a test tube, the lymphocytes are perfectly capable of killing tumor cells. But in the body, for some reason, they can't seem to stop a lesion from growing. So for melanoma, some researchers are working with a cream that can increase a tumor's "foreignness" to the immune system, tagging it to look more like an unwelcome virus and less like a familiar self cell. Other groups are testing ways to shut off the immune suppressors that the tumor sends out to hinder the natural seek-and-destroy tendencies of the immune system. That makes sense. Supercharging the immune system while the immune suppressors are still at work is a little like revving a car engine without releasing the emergency brake: in both cases, you're not going anywhere. And yet most early vaccine efforts have involved stepping on the gas alone.
One other way to get the immune system moving might be, in effect, to replace it with an entirely new one, says Rosenberg. If a vaccine can marshal the body's defenses to recognize and destroy a tumor, could you rebuild those defenses from the ground up and this time design them so they'll be especially good at fighting cancer cells?
Rosenberg's thinking is based on the now familiar strategy of the bone-marrow transplant for leukemia and lymphoma, which are blood- and immune-cell cancers. Radiation is used to obliterate a patient's cancer-tainted immune cells; those cells are then replaced by a population of new ones harvested from a healthy donor or grown from some of the patient's healthy cells. Rosenberg refines this method for melanoma by first exposing immune-system cells to tumor cells in a dish, thus "training" them to sprout proteins that target cancer cells, and only then infusing them into patients. Already he has shown that such a fortified mix can cause tumor regression in up to 70% of melanoma patients.
Even that, Rosenberg says, can be improved on. He is tipping the odds further in favor of the anticancer cells by genetically modifying the tumor-fighting T cells so that cancer cells aren't simply among the ones they recognize but are the only ones they recognize — eliminating the distraction of other infections and allowing the T cells to devote all their energy to the malignancy alone. In June he published results showing that such manipulation can cause regression of tumors in one-third of subjects. "I think the most important progress in using the immune system is not by a vaccine but by using cell-transfer approaches," says Rosenberg. "Those are looking to be far more effective."
Measuring that effectiveness will be another challenge. The melanoma- and lymphoma-vaccine studies both tracked only the extent to which tumors regressed and were not designed to document what most cancer experts — not to mention patients — see as the gold standard of any new therapy: survival. Do patients who are vaccinated live longer than those who are not? How do the vaccine's cancer-controlling powers compare with those of the expanding list of drugs designed to sneak in and halt growing lesions by shutting off their supply of nutrients and oxygen or hampering their growth spurts?
Solving those riddles might be the most formidable challenge yet for the vaccine field. Some experts are already questioning the need for the lymphoma vaccine when a drug, rituximab, exists to control the disease. Kwak points out, however, that in addition to being able to seek out small deposits of tumor cells that even the best-targeted drug therapies might miss, vaccines are generally less toxic. Rituximab, for instance, can lead to viral infections and heart problems and may be toxic to the kidneys. If, as some researchers hope, cancer is ever to become more of a chronic disease like diabetes, which can be managed for life, finding treatments that are safe and effective over many years becomes critical. "The risk-benefit ratio begins to swing more against chemotherapy or targeted agents for long-term maintenance," says Kwak. "Whereas a vaccine, with a favorable safety profile, is ideal for that kind of setting."
If that's true, then this first group of cancer vaccines is well on its way to seeding an entirely new field of immune-based treatments for cancer. "In some way, shape or form, our body repairs cancer cells and 'prevents' cancer," says Lichtenfeld. "If it didn't, we would have much more cancer than we actually see. How simple it would be for us to take some markers on a cancer cell's surface and create a vaccine to help the body do what it's supposed to do." It's not simple at all, as it turns out, but it's an idea whose power and potential certainly make it worth the effort.
Deadly new flu strain erupts in Mexico, U.S.

By Alistair Bell and Noel Randewich
MEXICO CITY (Reuters) - A strain of flu never seen before has killed up to 60 people in Mexico and also appeared in the United States, where eight people were infected but recovered, health officials said on Friday.
Mexico's government said at least 20 people have died of the flu and it may also be responsible for 40 other deaths.
It shut down schools and canceled major public events in Mexico City to try to prevent more deaths in the sprawling, overcrowded capital. Authorities said they had enough antiviral medicine to treat about 1,000 suspected cases reported so far.
The World Health Organization said tests showed the virus from 12 of the Mexican patients was the same genetically as a new strain of swine flu, designated H1N1, seen in eight people in California and Texas.
"Our concern has grown as of yesterday," Dr. Richard Besser, acting director of the U.S. Centers for Disease Control and Prevention told reporters in a telephone briefing.
Global health officials were not ready to declare a pandemic -- a global epidemic of a new and deadly disease such as flu. "So far there has not been any change in the pandemic threat level," Besser said.
But the human-to-human spread of the new virus raised fears of a major outbreak. Mexico's government suspended classes for millions of children in Mexico City, where scared residents rushed out to buy face masks and kept their kids at home.
"We're frightened because they say it's not exactly flu, it's another kind of virus and we're not vaccinated," said Angeles Rivera, 34, a government worker who fetched her son from a public kindergarten that was closing.
Close analysis showed the disease is a mixture of swine, human and avian viruses, according to the CDC.
Humans can occasionally catch swine flu from pigs but rarely have they been known to pass it on to other people.
Mexico reported 1,004 suspected cases of the new virus, including four possible cases in Mexicali on the border with California.
Most of the dead were aged between 25 and 45, a health official said. It was a worrying sign as seasonal flu can be more deadly among the very young and the very old but a hallmark of pandemics is that they affect healthy young adults.
Health Minister Jose Angel Cordova said Mexico has enough antiviral drugs to combat the outbreak for the moment. "In the last 20 hours, fewer serious cases of this disease and fewer deaths have been reported," he told reporters.
The WHO said the virus appears to be susceptible to Roche AG's flu drug Tamiflu, also known as oseltamivir, but not to older flu drugs such as amantadine.
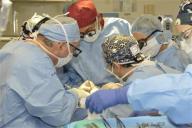
First U.S. patient gets face transplant
By Maggie Fox, Health and Science Editor
WASHINGTON (Reuters) - Surgeons have replaced 80 percent of a woman's face, transplanting bone, teeth, muscle and nerve in the first such operation in the United States.
They said the woman suffered severe trauma that cost her an eye, much of her nose and her upper jaw and left her unable to breathe, smell, taste or smile properly.
"You need a face to face the world," Dr. Maria Siemionow, director of plastic surgery research at the Cleveland Clinic in Ohio, where the operation was performed, told a news conference on Wednesday.
The clinic gave no details about the woman, how she was injured, or who the donor was.
The operation is the fourth done globally. French surgeons replaced much of the face of a woman in 2005 after she was disfigured in an attack by her dog. Last year, her doctors reported that she recovered slowly and steadily, overcoming two episodes of rejection.
In 2006, Chinese doctors performed a face transplant on a 30-year-old mauled by a bear, and a French team did a transplant in 2007 on a 29-year-old man who suffered from von Recklinghausen disease, which deforms the face.
The U.S. operation, which was performed two weeks ago, took 22 hours. A team of eight specialists transplanted bone, muscle, blood vessels and nerves.
"We transferred the skin, all the facial muscles in the upper face and mid-face, the upper lip, all of the nose, most of the sinuses around the nose, the upper jaw including the teeth, the facial nerve," said Francis Papay, chair of dermatology and plastic surgery at the clinic.
"Our hopes are ... that she will be able to smile again."
Some controversy has surrounded the issue of face transplants, in part because face injuries are not seen as life-threatening. In this case, the doctors said, the patient had virtually no life because of her injury.
"We know that there are so many patients there in their homes where they are hiding from society because they are afraid to walk to the grocery stores, they are afraid to go the the street," Siemionow said. "Our patient was called names and was humiliated."
"We very much hope that for this very special group of patients there is a hope that someday they will be able to go comfortably from their houses and enjoy the things we take for granted."
Dr. Eric Kodish, a bioethicist, said a team of experts, called an institutional review board, approved the operation.
"This is not a cosmetic surgery in any ... sense. The face is the embodiment of personal identity," Kodish told the news conference.
"A person who has sustained trauma or other devastation to the face is generally isolated and suffers tremendously," Kodish added. "We have hope that our patient will begin to smile again, will be able to smell again." Continued...
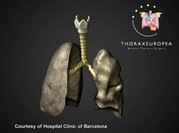
Doctors transplant windpipe with stem
By MARIA CHENG, AP Medical Writer Maria Cheng, Ap Medical Writer – 34 mins ago
LONDON – Doctors have given a woman a new windpipe with tissue grown from her own stem cells, eliminating the need for anti-rejection drugs. "This technique has great promise," said Dr. Eric Genden, who did a similar transplant in 2005 at Mount Sinai Hospital in New York. That operation used both donor and recipient tissue. Only a handful of windpipe, or trachea, transplants have ever been done.
If successful, the procedure could become a new standard of treatment, said Genden, who was not involved in the research.
The results were published online Wednesday in the medical journal, The Lancet.
The transplant was given to Claudia Castillo, a 30-year-old Colombian mother of two living in Barcelona, suffered from tuberculosis for years. After a severe collapse of her left lung in March, Castillo needed regular hospital visits to clear her airways and was unable to take care of her children.
Doctors initially thought the only solution was to remove the entire left lung. But Dr. Paolo Macchiarini, head of thoracic surgery at Barcelona's Hospital Clinic, proposed a windpipe transplant instead.
Once doctors had a donor windpipe, scientists at Italy's University of Padua stripped off all its cells, leaving only a tube of connective tissue.
Meanwhile, doctors at the University of Bristol took a sample of Castillo's bone marrow from her hip. They used the bone marrow's stem cells to create millions of cartilage and tissue cells to cover and line the windpipe.
Experts at the University of Milan then used a device to put the new cartilage and tissue onto the windpipe. The new windpipe was transplanted into Castillo in June.
"They have created a functional, biological structure that can't be rejected," said Dr. Allan Kirk of the American Society of Transplantation. "It's an important advance, but constructing an entire organ is still a long way off."
So far, Castillo has shown no signs of rejection and is not taking any immune-suppressing drugs, which can cause side effects like high blood pressure, kidney failure and cancer.
"I was scared at the beginning," Castillo said in a press statement. "I am now enjoying life and am very happy that my illness has been cured."
Her doctors say she is now able to take care of her children, and can walk reasonable distances without becoming out of breath. Castillo even reported dancing all night at a club in Barcelona recently.
Genden said that Castillo's progress needed to be closely monitored. "Time will tell if this lasts," he said. Genden added that it can take up to three years to know if the windpipe's cartilage structure is solid and won't fall apart.
People who might benefit include children born with defective airways, people with scars or tumours in their windpipes, and those with collapsed windpipes.
Martin Birchall, who grew Castillo's cells at the University of Bristol, said that the technique might even be adapted to other organs.
"Patients engineering their own tissues is the key way forward," said Dr. Patrick Warnke, a surgeon at the University of Kiel in Germany. Warnke is also growing patients' tissues from stem cells for transplants.
Warnke predicted that doctors might one day be able to produce organs in the laboratory from patients' own stem cells. "That is still years away, but we need pioneering approaches like this to solve the problem," he said.
On the Net: http://www.lancet.com
Study: Large waist almost doubles premature death risk
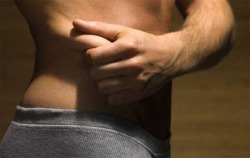
WASHINGTON, Nov. 12 (Xinhua) -- Having a large waistline can almost double your risk of dying prematurely even if your body mass index is within the "normal" range, according to a new study of over 350,000 people across Europe, published Wednesday in the U.S. magazine New England Journal of Medicine. The study provides strong evidence that storing excess fat around the waist poses a significant health risk, even in people not considered to be overweight or obese. It suggests that doctors should measure a patient's waistline and their hips as well as their body mass index as part of standard health checks, according to the researchers, from Imperial College London, the German Institute of Human Nutrition, and other research institutions across Europe. Comparing subjects with the same body mass index, the risk of premature death increased in a linear fashion as the waist circumference increased. The risk of premature death was around double for subjects with a larger waist (more than 120 cm for men and more than 100 cm for women) compared to subjects with a smaller waist (less than 80 cm for men and less than 65 cm for women). Body mass index is commonly used to assess if a person is of "normal" weight. Each 5 cm increase in waist circumference increased the mortality risk by 17 percent in men and 13 percent in women, according to the study. An increased risk of mortality may be particularly related to storing fat around the waistline because fatty tissue in this areasecretes cytokines, hormones and metabolically active compounds that can contribute to the development of chronic diseases, particularly cardiovascular diseases and cancers, suggest the authors. Tobias Pischon, the lead author of the paper, said "The most important result of our study is the finding that not just being overweight, but also the distribution of body fat, affects the risk of premature death of each individual."
From:www.chinaview.cn 2008-11-13 06:35:09
